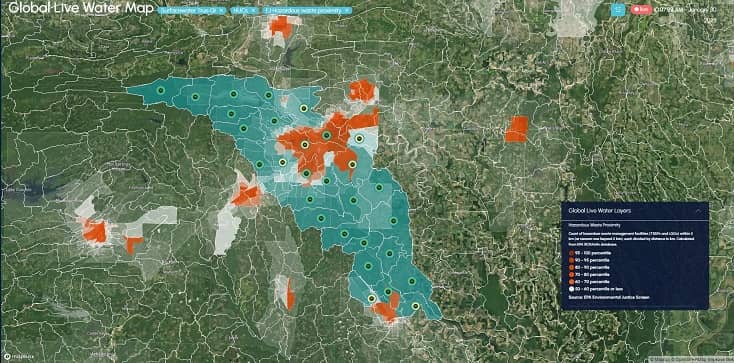
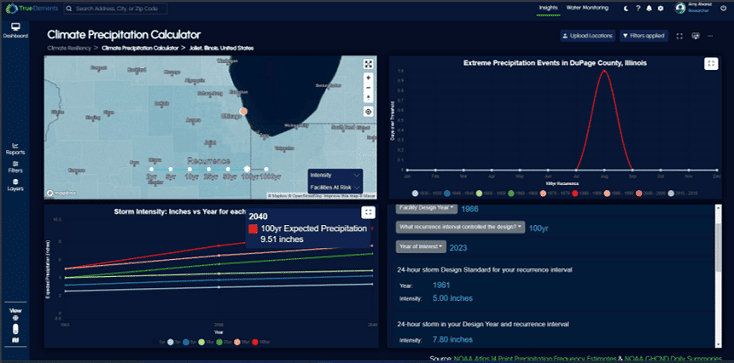
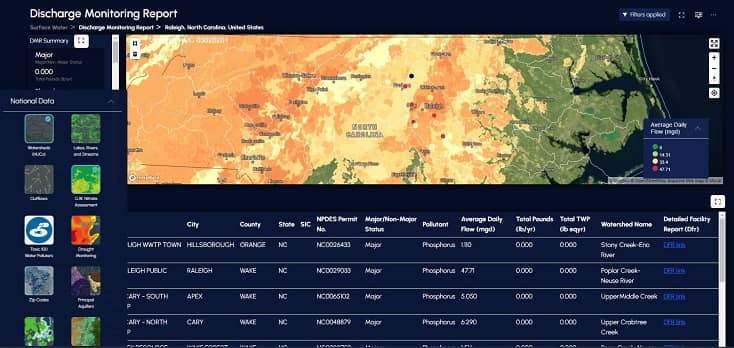
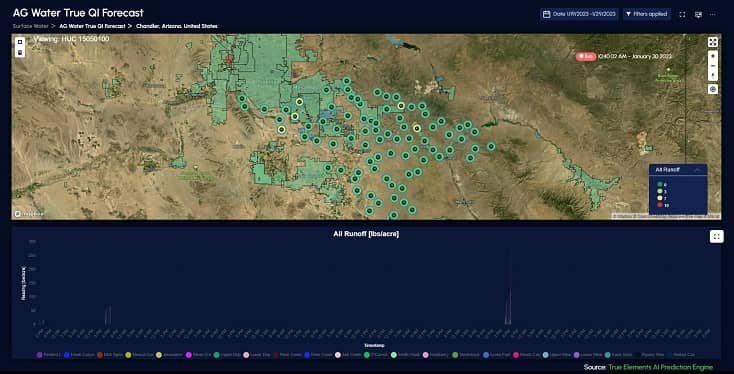
Partners Organizations Helping Us Deliver Water Intelligence
for a Secure Water Future
What Is Water Intelligence?
Water Intelligence is a unique combination of state-of-the-art technology, scientific analysis and AI capabilities that continuously aggregates, synthesizes, and transforms expansive amounts of complex water data into easily understandable, increasingly precise analyses and forecasts via scores and visualizations.
These analyses and forecasts produce deep, clear insights that help decision makers better understand water resources and make fully informed decisions about how to manage and optimize those resources.

Your Depends on Water.
Navigate Your Water Future Intelligently.
on water
The future of every business, community, government entity and industry depends on its ability to secure and maintain reliable, safe water resources.
Water is
complex
Water crises are accelerating
are shared
Water Resilience Starts With Water Intelligence.
True Elements is Your Dedicated Partner Providing the Water Intelligence You Need for the Insights You Want
- Watershed Health & Impacts
- Risk Assessment
- Regulatory Compliance
- Data Aggregation, Analysis & Forecasting
- Project Management & Data Sharing
When You Need Deeper Understanding Of:
Current and future water quantity issues
Current and future water quality issues
Interrelated watershed impacts
For informed site management and selection, supply chain challenge solutions, ESG initiatives, license to operate and reputational risk concerns.
When You Need Greater Awareness Of:
Current asset vulnerability
Key considerations for capex investments
Supply chain vulnerabilities
When You Need Clear Understanding Of:
Current and future water quality issues
Current and future water quantity issues
Interrelated watershed impacts
When You Need Scientific and AI Capabilities That:
Aggregate, normalize, and index data in a universal water data platform
Allow customized data integration from private sources
Provide continuous water quality and quantity monitoring
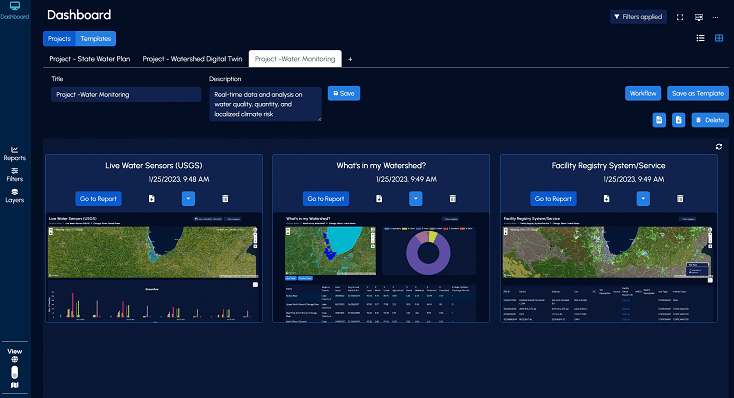
Provide an easy-to-use dashboard to holistically manage water challenges
When You Need Capabilities That Facilitate Collective Action:
Ability to incorporate site specific data from company or other private data sources
Customizable project dashboards and tools for project management and participation monitoring
Data download capability and API access to dashboards
Secure firewalls to separate access to data as necessary
Products work separately for specific needs or together to provide more holistic water intelligence.
What Industry Leaders Are Saying
In The News

SkyFi Announces Partnership with True Elements to Integrate Advanced Water Intelligence into its Platform

Innovations in Water Quality Monitoring

Big Data, Deeper Insights: Crafting Smarter Water Strategies
True Elements Provides The Deep, Clear Water Intelligence You Need For the Insights You Want
Transforming water and data complexity into clear, easy to understand insights for fully informed, effective decision making.